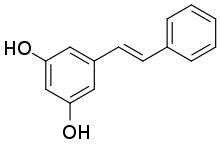
Pinosylvin
|
|
| Names | |
|---|---|
|
Preferred IUPAC name
5-[(1E)-2-Phenylethen-1-yl]benzene-1,3-diol |
|
| Other names
(E)-3,5-Stilbenediol
trans-3,5-Dihydroxystilbene |
|
| Identifiers | |
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.208.695 |
|
PubChem CID
|
|
| UNII | |
|
CompTox Dashboard (EPA)
|
|
|
|
|
|
| Properties | |
| C14H12O2 | |
| Molar mass | 212.248 g·mol−1 |
| Appearance | white solid |
| Melting point | 153 to 155 °C (307 to 311 °F; 426 to 428 K) |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
|
|
Pinosylvin is an organic compound with the formula C6H5CH=CHC6H3(OH)2. A white solid, it is related to trans-stilbene, but with two hydroxy groups on one of the phenyl substituents. It is very soluble in many organic solvents, such as acetone.
Occurrence
Pinosylvin is produced in plants in response to fungal infections, ozone-induced stress, and physical damage for example. It is a fungitoxin protecting the wood from fungal infection. It is present in the heartwood of Pinaceae and also found in Gnetum cleistostachyum.
Injected in rats, pinosylvin undergoes rapid glucuronidation and a poor bioavailability.
Biosynthesis
Pinosylvin synthase, an enzyme, catalyzes the biosynthesis of pinosylvin from malonyl-CoA and cinnamoyl-CoA:
- 3 malonyl-S-CoA + cinnamoyl-S-CoA → 4 CoA-SH + pinosylvin + 4 CO2
This biosynthesis is noteworthy because plant biosyntheses employing cinnamic acid as a starting point are rare compared to the more common use of p-coumaric acid. Two other compounds produced from cinnamic acid are anigorufone and curcumin.