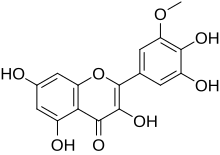
Laricitrin
|
|
| Names | |
|---|---|
|
IUPAC name
3,3′,4′,5,7-Pentahydroxy-5′-methoxyflavone
|
|
|
Systematic IUPAC name
2-(3,4-Dihydroxy-5-methoxyphenyl)-3,5,7-trihydroxy-4H-1-benzopyran-4-one |
|
| Other names
3'-O-Methylmyricetin
|
|
| Identifiers | |
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
|
PubChem CID
|
|
| UNII | |
|
CompTox Dashboard (EPA)
|
|
|
|
|
|
| Properties | |
| C16H12O8 | |
| Molar mass | 332.264 g·mol−1 |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
|
|
Laricitrin is an O-methylated flavonol, a type of flavonoid. It is found in red grape (absent in white grape) and in Vaccinium uliginosum (bog billberries). It is one of the phenolic compounds present in wine.
Metabolism
Laricitrin is formed from myricetin by the action of the enzyme myricetin O-methyltransferase. It is further methylated by laricitrin 5'-O-methyltransferase into syringetin.
Glycosides
- Laricitrin 3-O-galactoside, found in grape
- Laricitrin 3-glucoside found in Larix sibirica
- Laricitrin 3,5’-di-O-β-glucopyranoside, found in Medicago littoralis